 Typically, metals are both malleable and ductile. The distance also indicates the size of the moment, which is the same reason the compound is polar.Hint: The size of the P atom is greater than the N atom, down the group basicity of the atom decreases when the size of the atom increases and the electron density decreases. Study with Quizlet and memorize flashcards containing terms like The molecule with the largest dipole moment A CO2 B H2O C CH4 D C2H4 E PH3, A particle-level diagram of a metallic element is shown above. 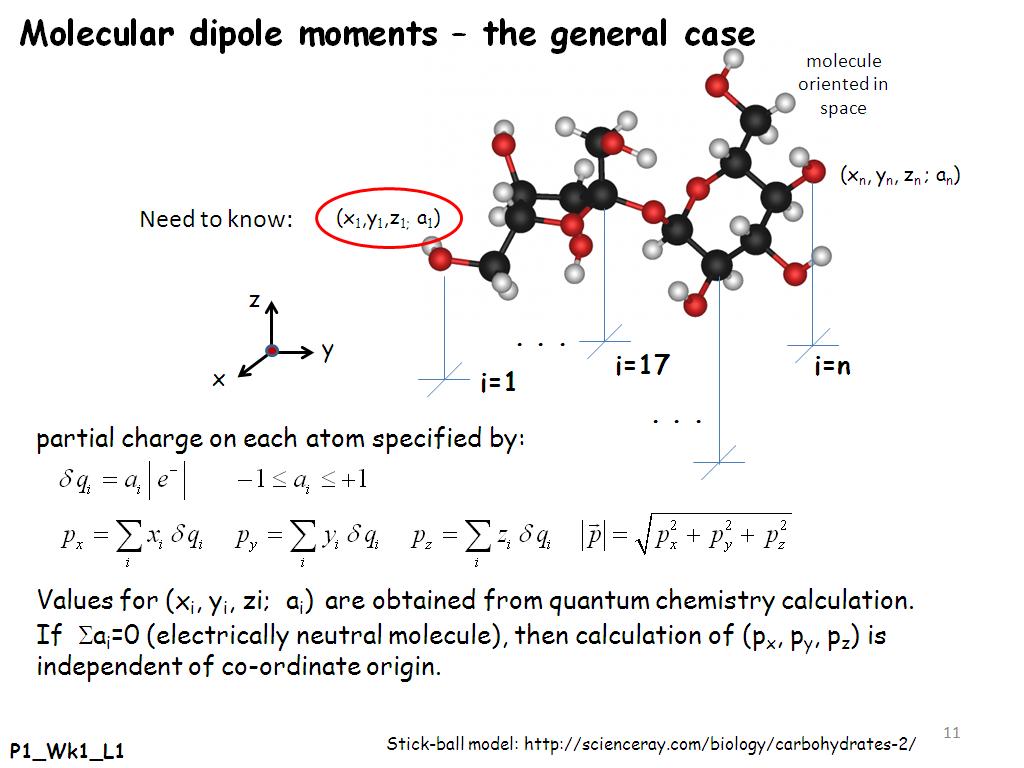
The more significant the electronegativity difference produces, the larger the moments. When atoms form polar bonds, the difference in electronegativity gives dipole. Because dipoles of the bond cancel each other. 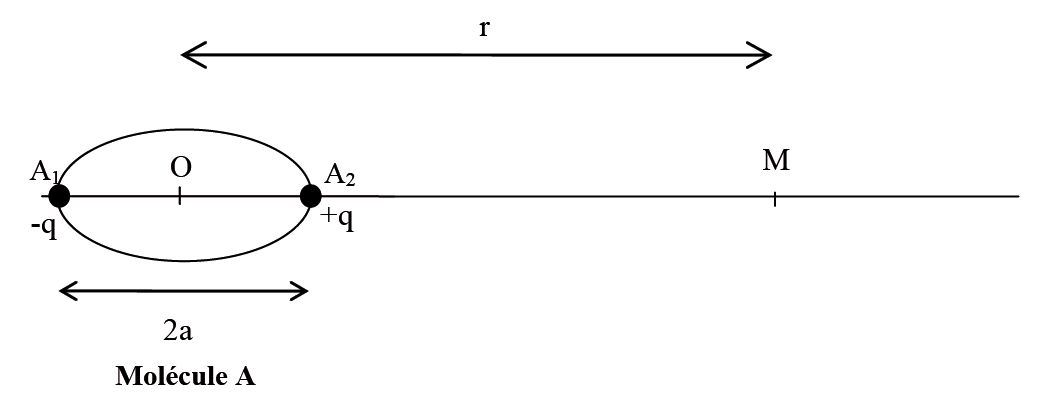
(i) (Refer to Image 1) (ii) (Refer to Image 2) (iii) (Refer to Image 3) (iv) (Refer to Image 4) Above molecules have dipole moment zero. When a compound creates Dipole moments, it occurs when there is a separation of charge. Dipole moment:- The polarity of covalent bond can be conveniently measured in terms of physical quantity called dipole moment. As the compound shares electrons unequally, a dipole moment occurs. It is essential to identify the dipole moment of the compound. The phosphine molecules have a dipole moment of 0.58D, much smaller than the NH3 dipole moment. These forces are more potent than the Van der Waals forces. With this, the PH3 is a polar molecule with nonpolar covalent bonds, not polar bonds. The molecules of the interhalogen compound PH3 form a dipole-dipole interaction and a hydrogen bond. Its use as an example here is both wrong and deeply misleading. methanol or water) IT is not correct to describe carbon dioxide as 'polar' in almost any sense in chemistry. Even though, CHX2ClX2 C H X 2 C l X 2 is rather somewhat polar than polar ( like e.g. However, due to the unbonded electron, there will be asymmetrical charge distribution. CHX4 C H X 4 has dipole zero moment due molecule symmetry. 
The hydrides of group 5A are NH3, PH3, AsH3, and SbH3. Step 3: The boiling point of a compound increases with an increase in molar mass. Highest to lowest - SbH3, AsH3, PH3, and NH3. Be sure to label the indicate: 1.Dipole moment (arrow) of each bond 2.Net dipole 3.Partial positive and partial negative 4.Molecular shape. Question: Draw the Lewis structure of H2O and PH3. The electronegative property of PH3 found in the periodic table attracts shared pairs of valence electrons, creating covalent bonds. Arrange the group 5A hydrides PH3, SbH3, NH3, and AsH3 from highest to lowest molar mass. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. 7.99d How to determine if PCl3 has a dipole moment. 
In one lone pair orbital with three hydrogen bonds, Hydrogen atoms and a Phosphorus atom have the same electronegativity. shorts bond angle between PH3,PF3,PCl3 PBr3,PI3 Veena Dixit Chemistry IIT jee. Phosphine or Phosphorus Trihydrate is one best candidate for being polar molecules that have nonpolar bonds. The molecules of the interhalogen compound PH3 form a dipole-dipole interaction and a hydrogen bond. Determine whether each compound below exhibits a molecular dipole moment: (a) CH4 (b) PH3 (c) H2O (d) CO2 (e) CCl4 (f) CH2Cl2 EXPLAIN. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
